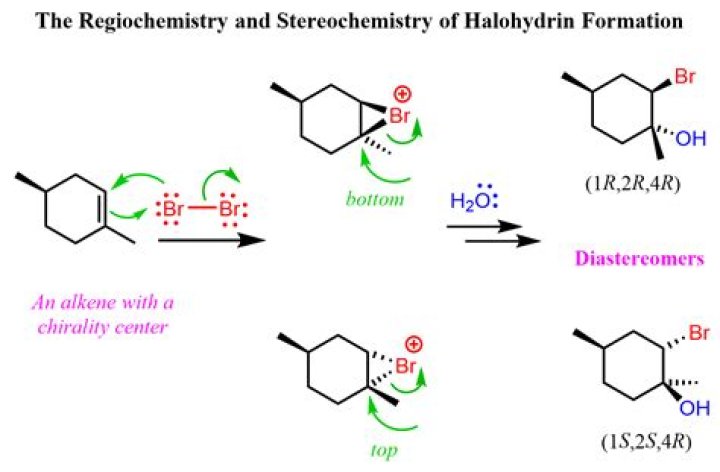
Alkenes are generally prepared through β elimination reactions, in which two atoms on adjacent carbon atoms are removed, resulting in the formation of a double bond. Preparations include the dehydration of alcohols, the dehydrohalogenation of alkyl halides, and the dehalogenation of alkanes.
How are alkenes formed from alkanes?
An alkene represents an unsaturated hydrocarbon with double bonds, while an alkane is a saturated hydrocarbon with only single bonds. To convert an alkane to an alkene, requires that you remove hydrogen from the alkane molecule at extremely high temperatures. This process is known as dehydrogenation.
How are alkynes formed?
The loss of a hydrogen atom and a halogen atom from adjacent alkane carbon atoms leads to the formation of an alkene. The loss of additional hydrogen and halogen atoms from the double‐bonded carbon atoms leads to alkyne formation.
Which alkene is not Colourless?
Which among the following is not colourless? Explanation: Methene compound does not exist according to the formula CnH2n and also due to the lack of C=C. 8.
How do you make an alkene from an alcohol?
The dehydration reaction of alcohols to generate alkene proceeds by heating the alcohols in the presence of a strong acid, such as sulfuric or phosphoric acid, at high temperatures.
16 related questions foundWhat is hydrogenation of alkenes?
An example of an alkene addition reaction is a process called hydrogenation.In a hydrogenation reaction, two hydrogen atoms are added across the double bond of an alkene, resulting in a saturated alkane.
How are alkenes prepared from alkyl halides?
From alkyl halides: Alkenes are obtained by heating alkyl halides with alcoholic potash. Alcoholic potash is obtained by dissolving potassium hydroxide in alcohol. In this reaction, dehydrohalogenation takes place i.e. a single molecule of halogen acid is removed.
How alkenes are produced using fractional distillation?
Commonly, after fractionation, long chain alkanes are passed over a hot catalyst in a process called cracking. During this process, the long alkanes are broken up into short alkanes and alkenes. Alkenes have a double bond, which means they can be turned into polymers to make plastics, or alcohols and other derivatives.
Are alkenes formed in cracking?
Cracking is important for two main reasons: It helps to match the supply of fractions with the demand for them. It produces alkenes, which are useful as feedstock for the petrochemical industry.
Why are alkenes so important?
In manufacturing, alkenes find many different applications. They are used in the synthesis of alcohols, plastics, lacquers, detergents, and fuels as starting materials. For the chemical industry, the most important alkenes are ethene, propene, and 1,3-butadiene.
What are two methods of cracking?
Cracking is primarily of two types – thermal cracking and catalytic cracking.
How is alkene produced by Kolbe's electrolytic method?
Sodium or potassium salt of a dicarboxylic acid on electrolysis gives an alkene. An alkene is generated when an aqueous solution of sodium or potassium salt of a dibasic acid (with adjacent carboyxlic groups) is electrolyzed.
How alkenes and alkynes are prepared?
Alkenes can be prepared from alkynes by carrying out hydrogenation in the presence of palletised charcoal. The charcoal which is used in this reaction has been moderately deactivated. Lindlar catalyst is palladium on calcium carbonate which has been deactivated by lead acetate to stop further hydrogenation.
How is alkene produced by vicinal dihalides?
1 Answer. Vicinal dialyses on treatment with Zn metal lose a molecule of ZnX2 to from an alkene. This reaction is known as dehalogenation.
Why is alkene formation exothermic?
A majority of these reactions are exothermic, due to the fact that the C-C pi-bond is relatively weak (ca. 63 kcal/mole) relative to the sigma-bonds formed to the atoms or groups of the reagent.
Do alkenes react with H2?
Ch 6 : Alkenes + H2. Alkenes can be reduced to alkanes with H2 in the presence of metal catalysts such as Pt, Pd, Ni or Rh. The two new C-H σ bonds are formed simultaneously from H atoms absorbed into the metal surface. The reaction is stereospecific giving only the syn addition product.
What happens when hydrogen is added to alkenes?
When alkenes react with hydrogen gas in the presence of a variety of metal catalysts, a hydrogen molecule will be added to the double bond in the way that each carbon atom bonded with one hydrogen atom, such addition reaction is called hydrogenation.
What is the chemical reaction of alkenes?
The most common type of reaction for alkene is the addition reaction to C=C double bond. In addition reaction, a small molecule is added to multiple bond and one π bond is converted to two σ bonds (unsaturation degree decreases) as a result of addition. Addition reaction is the opposite process to elimination.
What does Markovnikov's rule state?
Markovnikov rule, in organic chemistry, a generalization, formulated by Vladimir Vasilyevich Markovnikov in 1869, stating that in addition reactions to unsymmetrical alkenes, the electron-rich component of the reagent adds to the carbon atom with fewer hydrogen atoms bonded to it, while the electron-deficient component ...
What are the three methods of preparation of alkanes and alkenes?
General Methods of Preparation of Alkanes
- Decarboxylation.
- Wurtz Reaction.
- By the Reduction of Alkyl Halides.
- By Hydrogenation of Alkenes((>C=C<) : Sabatier and Senderen's Method.
- Kolbe's Electrolysis Method.
- By Grignard Reagents.
- By Reduction of Alcohols, Aldehydes, Ketones or Fatty Acids and their Derivatives.
Which of the following alkenes can be produced by Kolbe electrolysis?
Therefore, the required answer is (iii) Ethane (CH3−CH3) .
How do you make an alkane?
Alkanes can be prepared by reduction of haloalkanes. One of these preparation methods of alkanes was given by Wurtz which is called Wurtz Reaction. When alkyl halide reacts with metallic sodium in presence of dry ether, it forms alkane with double the number of carbon atoms present in the alkyl halide of reactants.
What is the electrolysis product of sodium succinate?
By the electrolysis of aqueous sodium succinate, ethene is formed at along with .
How do you test for alkenes?
A simple test with bromine water can be used to tell the difference between an alkane and an alkene. An alkene will turn brown bromine water colourless as the bromine reacts with the carbon-carbon double bond. In fact this reaction will occur for unsaturated compounds containing carbon-carbon double bonds.
Are alkenes saturated?
The alkenes are unsaturated. This means that they have a carbon to carbon double bond. The alkanes are saturated because they only contain single bonds.