Sugar lowers the freezing point of water, which makes frozen desserts fair game for changes in freezing point.
Does sugar keep ice from melting?
This is because in the same amount or volume, there are more molecules of salt than sugar or baking soda due to the chemical make-up. Salt, baking soda, and sugar will all act to lower the freezing point of the ice, making it melt quicker than the untouched ice cube.
Does sugar freeze ice faster?
Still partly a liquid. We concluded that different substances can make a difference in how fast water freezes. The sugar water froze the fastest.
How does sugar affect the freezing point of ice cream?
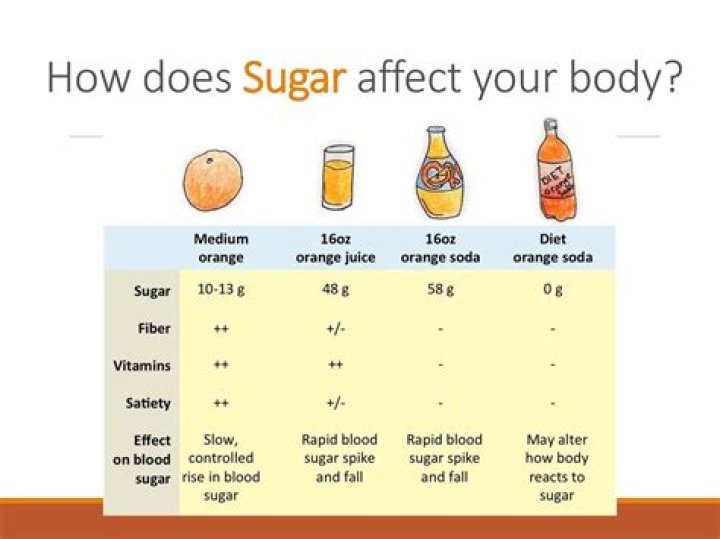
Sugar affects the freezing point of foods. The higher the concentration of sugar, the lower the freezing point. A low freezing point is important in ice cream and frozen desserts. It reduces the risk of large crystals of ice forming.
What is freezing point of sugar?
The melting/freezing temperature of a common sugar (sucrose) is 185 °C or 365 °F.
15 related questions foundWhat slows ice melting?
General insulation of the ice causes it to melt slower. Wrapping it in wool, Styrofoam or wood contains the cold air emitting from the ice, keeping the temperature of the ice low. Putting the ice in a vacuum, such as a vacuum-insulated Thermos bottle, also prevents the ice from melting quickly.
Why do you think the sugar affects the freezing time of water?
Water freezes at 32 degrees Fahrenheit (0 degrees Celsius), but when a solute such as sugar is added, the freezing point changes. The sugar molecules prevent the water from making hydrogen bonds, which are required for solidity, and the water has to become even colder before it reaches its freezing point.
What happens when sugar melts?
When simple sugars such as sucrose (or table sugar) are heated, they melt and break down into glucose and fructose, two other forms of sugar. Continuing to heat the sugar at high temperature causes these sugars to lose water and react with each other producing many different types of compounds.
Does sugar melt or decompose?
Sugar crystals do not melt, but instead decompose in a heat sensitive reaction termed 'apparent melting', according to new research. The research, published in the Journal of Agricultural and Food Chemistry and recently presented to the Institute of Food Technologists, goes against years of belief that sugar melts.
Does sugar dissolve or melt?
Students consider that sugar melts when it dissolves in water. Often melting is considered to be substances turning into water. This is further reinforced because children see heat being involved in both processes - they know if you want to dissolve more sugar you warm up the water.
Why does sugar melt easily?
Hence, its melting point is very high. Sucrose is a covalent compound and no ions are present. Molecules are held together by weak van der Waal forces and hydrogen bonds. Hence, it has low melting point.
What happens if you freeze sugar water?
Frozen water (ice) is a regular array of molecules. Sugar molecules don't fit well in that array. So when you freeze sugar water or juice, the sugar stays behind in the liquid water as the ice forms.
What does salt do to ice?
Why does salt melt ice and snow? Salt causes a phenomenon called freezing point depression, which means it lowers the freezing point of water. Water normally freezes at 32 degrees Fahrenheit, but when you add salt, that threshold can drop as low as 20 degrees Fahrenheit.
What can make ice last longer?
8 Tips for Keeping Ice Longer in Your Icebox
- Prepare your icebox. ...
- Use block ice. ...
- Add some salt to your ice. ...
- Cool your drinks and food first. ...
- Adding the beers! ...
- Keep it out of the sun. ...
- Avoid opening your icebox too much. ...
- Fill your icebox as much as you can.
Does salt melt ice faster?
Because salt particles make it harder for water particles to freeze back onto the ice, the ice that is in contact with dissolved salt melts faster. When the saltwater flows over the surface it melts the ice on its way, creating channels, like rivers, over the surface of the ice ball.
How do you make ice cubes not melt?
Place a towel in the bucket or container before putting in the ice cubes. This will insulate the ice and keep it cold. You can also use bubble wrap and then a towel in the container to keep the ice insulated and less prone to melting.
Does putting salt on ice make it colder?
When water freezes from being in cold air, the release of heat actually slows down the freezing. When you add salt to a mixture of water and ice, it causes more ice to melt by depressing the freezing point and not by adding internal energy so it actually gets colder.
Will Epsom salt melt ice?
As the regular table salt, Epsom salt also melts the ice but in a very slow manner and is also costlier than regular salt. Epsom salt, also called Magnesium Sulfate, can be used for melting ice in the same process as normal salt. The freezing point of ice is lowered and thereby melting the ice or snow.
Can you put table salt on ice?
1. Table salt. Instead of rock salt, you can sprinkle a thin layer of table salt over icy areas. Heat is generated because of the chemical reaction that takes place between the salt and water, which lowers the freezing point of the water in the snow.
What is the behavior of sugar before melting?
The chemical reaction we are most familiar with is that of melting: sugar decomposes at a temperature ranging between 184 and 186°C. This is a very recent discovery we owe to a team of researchers in Illinois. Basically, when we heat sucrose gently, this produces a phenomenon known as “apparent melting”.
What is melted sugar called?
Caramelizing sugar is a term most often applied to melting sugar until it becomes a caramel color liquid. Caramelized sugar is simply a mixture of sugar and water cooked until it becomes syrupy and darkens, and reaching a temperature from 340 to 350 degrees F.
How hot does melted sugar get?
Because the process of making sugar involves heating and cooling it, sugar manufacturers have learned that the melting point of sucrose (table sugar) is 367 degrees F. Knowing this information, we can use sugar to test the accuracy of your oven's temperature.
Can sugar melt in cold water?
Sugar dissolves faster in hot water than it does in cold water because hot water has more energy than cold water.
How long does it take sugar to dissolve in cold?
Sugar dissolves in water at 0 degrees Celsius. This is because sugar is soluble in liquid water at all temperatures at standard pressure. It takes 10min. to dissolve in cold water, with occasional stirring.
Does sugar ionize in water?
Since it does not "dissociate," I would say that the answer is no, it does not "ionize," and therefore has a Van't Hoff factor of 1 when mixed with pure water. Like anything with electrons, they can be lost.